The Trump administration filed an appeal to Fourth Circuit Court of Appeals to reinstate an FDA rule that requires women to visit a hospital, clinic or medical office to obtain a chemical abortion during the Coronavirus.
Obama-appointed U.S. District Judge Theodore Chuang ruled to temporarily block enforcement of the FDA rule requiring the abortion pill to be administered only in clinics, medical offices or hospitals. Chuang’s ruling allowed the abortion pill to be mailed to women without seeing a doctor.
The Justice Department asked Judge Chuang to suspend his July 13 order pending the appeal, but the judge refused.
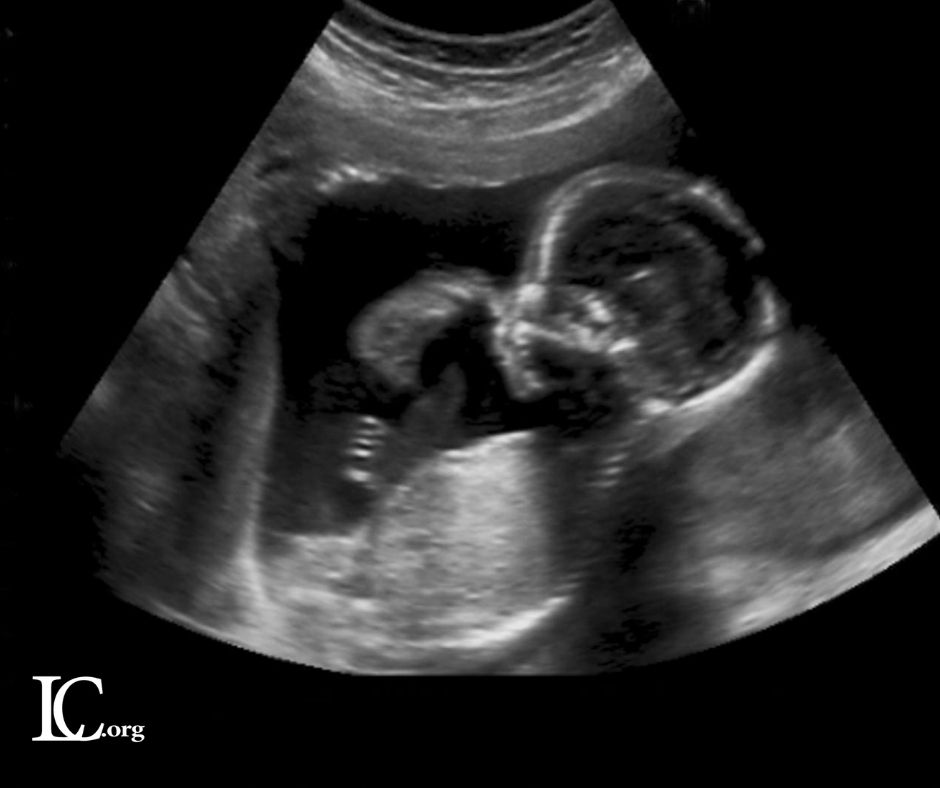
The judge’s ruling will allow health care providers to arrange for abortion pills to be mailed or delivered to patients during the public health emergency declared by the secretary of the U.S. Department of Health and Human Services. The U.S. Food and Drug Administration approved mifepristone to be used in combination with a second drug, misoprostol, to end an early pregnancy or manage a miscarriage. Mifepristone can be administered up to 10 weeks into a pregnancy when the baby has a beating heart and arms and legs. The first pill in the regimen, mifepristone, cuts off blood and nutrients to the baby, slowly starving it to death over one to two days. The second pill induces labor.
Chuang concluded that the “in-person requirements” for patients seeking abortion drugs impose a “substantial obstacle” to abortion patients and are likely unconstitutional under the circumstances of the pandemic.
he Justice Department wrote in its appeal, “Defendants will … suffer irreparable harm in the absence of a stay because they will be unable to enforce requirements that FDA has determined, based on its experience and scientific expertise, are necessary to ensure safe use of Mifeprex [or, mifepristone]. Requiring patients to obtain Mifeprex at a clinic – as has been required for years – does not deprive women of the ‘ability to make a decision to have an abortion.’”
The in-person dispensing requirement, the Justice Department said, “is not to monitor patients for complications, but rather to ensure that: (1) at the time of dispensing, the patient has the opportunity to receive counseling about the risk of serious complications associated with Mifeprex and what to do should they arise; and (2) there is no delay in the patient receiving their Mifeprex prescription, which could increase the risks of serious bleeding or infection.”
Chemical abortions account for approximately 40 percent of all abortions committed and the risk of death for the woman appears to be 10 times greater than from surgical abortions. A recent study published in the journal Frontiers in Neuroscience provides additional evidence of the harmful biological and behavioral effects of drug-induced abortion.
The FDA specifies notes that chemical abortion pills should never be given to a woman if she cannot go to a follow up visit to check on possible complications. As of December 31, 2018, there have been 24 reported deaths of women in the United States associated with mifepristone since U.S. Food and Drug Administration approved the drug in September 2000. This includes two cases of ectopic pregnancy resulting in death and eight fatal cases of severe systemic infection (also called sepsis). There have also been nearly 600 women experiencing such severe blood loss that they required transfusions.
Liberty Counsel Founder and Chairman Mat Staver said, “Abortion drugs are not safe. Abortion harms women and kills children.”